Vsepr model11/18/2023 The graphic in this article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. If you want to learn more about working out the shapes of molecules using VSEPR, check out the provided links in the ‘further reading’ section below. This leads to the range of different shapes for the same number of electron pairs, as indicated in the graphic. The VSEPR model is a model which predicts the geometrical shapes of molecules based. Consequently, if they are present in a molecule, they can have an effect on the shape, taking up a space that would usually be occupied by a bonding pair, and making the angles between the other bonds in the molecule marginally smaller (by approximately 2.5˚ per lone pair present around the central atom). The VSEPR model stands for Valence Shell Electron Pair Repulsion model. VSEPR Model- (valence shell electron pair repulsion model). The presence of electrons that are not involved in bonding around the central atom, so-called ‘lone pairs’, are closer to the central atom, and as such repel electron pairs around them more strongly. The overall shape of a molecule is determined by: Bond angles. The VSEPR model is not a theory it does not attempt to explain observations. 
Note that this method assumes that only single bonds are present in the molecule these are formed using a pair of shared electrons (1 from each atom), whereas double bonds involve two pairs of shared electrons.Ī complication of VSEPR is that all electrons do not repel equally. The VSEPR model can predict the structure of nearly any molecule or polyatomic ion in which the central atom is a nonmetal, as well as the structures of many molecules and polyatomic ions with a central metal atom. This can be determined in a few short steps, using the method provided in the graphic. The model states that electron pairs will repel each other such that the shape of. In order to work out the shape adopted, we simply need to find the number of electron pairs surrounding the central atom in a molecule. The acronym VSEPR stands for the valence-shell electron pair repulsion model. However, it generally gives good predictions for compounds of main group elements. It doesn’t take into account factors such as the size of bonded atoms or groups, and as such doesn’t always predict the shape of certain compounds correctly, in particular those of transition metals. Valence Shell Electron Pair Repulsion Theory ( VSEPR) is a molecular model to predict the geometry of the atoms making up a molecule where the electrostatic forces between a molecules valence electrons are minimized around a central atom. VSEPR works on the assumption that the shape adopted is that which minimises repulsions between the electron pairs in the molecule. She considers chemistry to be a bridge between biology and physics that can help explain observations made in everyday life.One for the chemistry students (and teachers!) out there today, with a look at how we can work out the shapes of some simple molecules using Valence Shell Electron Pair Repulsion (VSEPR) theory. These shapes are decided by the arrangement of electrons around the central atom in the molecule. The VSEPR model can predict the structure of nearly any molecule or polyatomic ion in which the central atom is a nonmetal, as well as the structures of many molecules and polyatomic ions with a 3. It is shown how recent developments in the analysis of electron. Exceptions to the model are discussed and are shown to have led to improvements in the model and to the development of another model the LCP model which supplements the VSEPR model. Stefanie Sydlik describes how an exciting “Take Your Daughter to Work Day” convinced her to become a chemist. The history and development of the VSEPR model since its inception in 1957 are reviewed. Stefanie’s sensor design amplifies small-scale changes in bond angles following interaction with a target molecule, and she hopes that her work will lead to better detectors for landmines and improvised explosive devices. What is VSEPR used in chemistry It is used to predict the molecular shape of molecules. Stefanie Sydlik explains how her research on designing sensors for explosives depends on the principles of VSEPR (valence shell electron pair repulsion) theory. The most important feature of the VSEPR model is the dominating influence of non-bonding or lone pairs of electrons in the valence shell of the central atom. 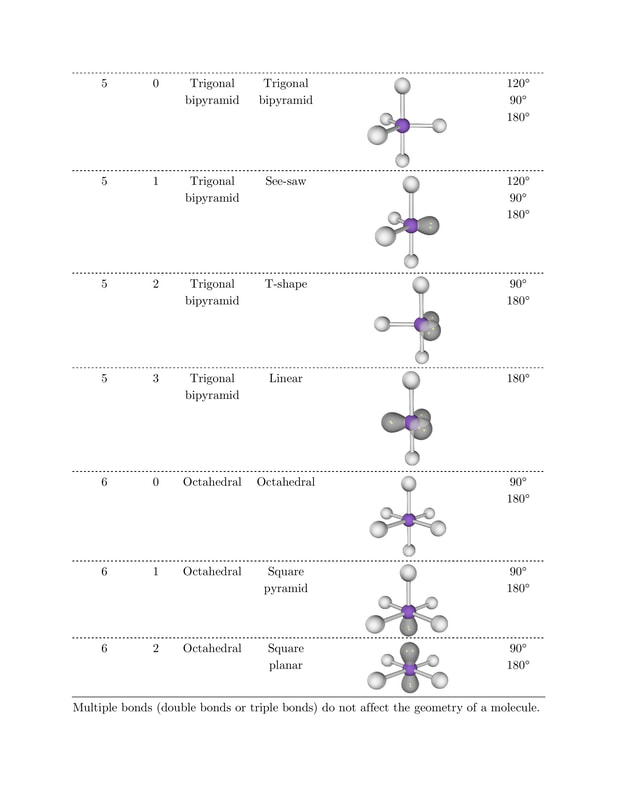
Molecules with Lone Pairs on the Central Atomĭetecting Explosives To Save Lives In War Zone Valence shell electron pair repulsion (VSEPR) theory is a model used in chemistry to predict the geometry of individual molecules from the number of. Lecture 12 Clicker Questions (PDF) Textbook Reading Notes for Lecture 12 (PDF) Clicker Questions
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
